|
"Atomic Weight: The Name, Its History, Definition and Units". Origin of the concept Jean Perrin in 1926 The Avogadro constant is named after the Italian scientist Amedeo Avogadro (17761856), who, in 1811, first proposed that the volume of a gas (at a given pressure and temperature) is proportional to the number of atoms or molecules regardless of the nature of the gas. For the output file that failed, I just tried opening the entire output file from Gaussian: a 5 MB. You’ll need to tweak the format once you paste, since Gaussian includes the atom index in the first column and charges (usually 0) in the 3rd column. In the SI dimensional analysis of measurement units, the dimension of the Avogadro constant is the reciprocal of amount of substance, N − 1 : Cite journal requires |journal= ( help) In Avogadro 1.9x it’s under Build > Atomic Coordinate Editor. 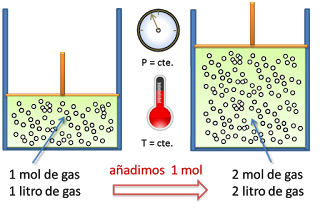
The constant is named after the physicist and chemist Amedeo Avogadro (1776–1856). Use Avogadros number to convert to moles and vice versa given the number of particles of an element. In practice, its value is often approximated as 6.02×10 23 mol -1 or 6.022×10 23 mol -1. I faced a minor issue while trying to adapt my workflow to the new version. If you had Avogadro installed in 18.04, and did a upgrade, it is replaced with Avogadro 2. Avogadro’s law, a statement that under the same conditions of temperature and pressure, equal volumes of different gases contain an equal number of molecules. 
Now the version available in the repositories is Avogadro 2. It is used as a normalization factor in the amount of substance in a sample (in SI units of moles), defined as the number of constituent particles (usually molecules, atoms, or ions) divided by N A. It seems Avogadro was phased out in Ubuntu 20.04 (Focal Fossa) and derivatives like Mint 20 (Ulyana). Then you know there is 2moles/2liters 1 mole per liter. And you know there is 2 moles of gas in this volume. We are actively porting more features to the Avogadro 2 code base, and making regular releases to get feedback from. The Avogadro 1.x series currently has more features, and can be found here. It is likely that, in 2015, Avogadro’s Number and Boltzmann’s constant will be given defined values. Because the number of particles is related to the number of moles (1 mol 6. This fundamental principle forms the basis of the ideal gas equation, a cornerstone in the. Avogadro 2 is being developed as part of the Open Chemistry project by an open community, and was started at Kitware as an open source community project. This quantity of electricity is generally called a faraday, and is about 96,484 coulombs, and is the product of the electronic charge and Avogadros number. Discover how the Italian chemist Amedeo Avogadros experiments with tiny particles led to the postulation that equal volumes of gas at the same temperature and pressure contain equal number of particles. An example: If you measure a containers volume is 2 liters. \begingroup What version of Avogadro2 do you have installed How did you install it How did you install it The latest version (1.95.1) fixes a number of packaging bugs related to input generators. Explore Avogadros Law with this engaging lesson. \( \newcommand\): Copy and Paste Caption here.The Avogadro constant, commonly denoted N A or L, is an SI defining constant with an exact value of 6.022 140 76 ×10 23 mol -1 ( unit of reciprocal moles). If you want to know the number of molecules per volume, you need to multiply this by Avogadros constant.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
